I Know I am Safe with
the Leading COVID-19
Home Test.
*Source: Circana Retail Sales Data (Units sold)
Expiration Date Extension
As of April 4, 2025, the expiration date of the Flowflex Plus COVID-19 and Flu A/B Home Test has been extended by 6 months. Please click here to check the new expiration date using the lot number printed on the test kit box
Simple. Fast. Convenient.
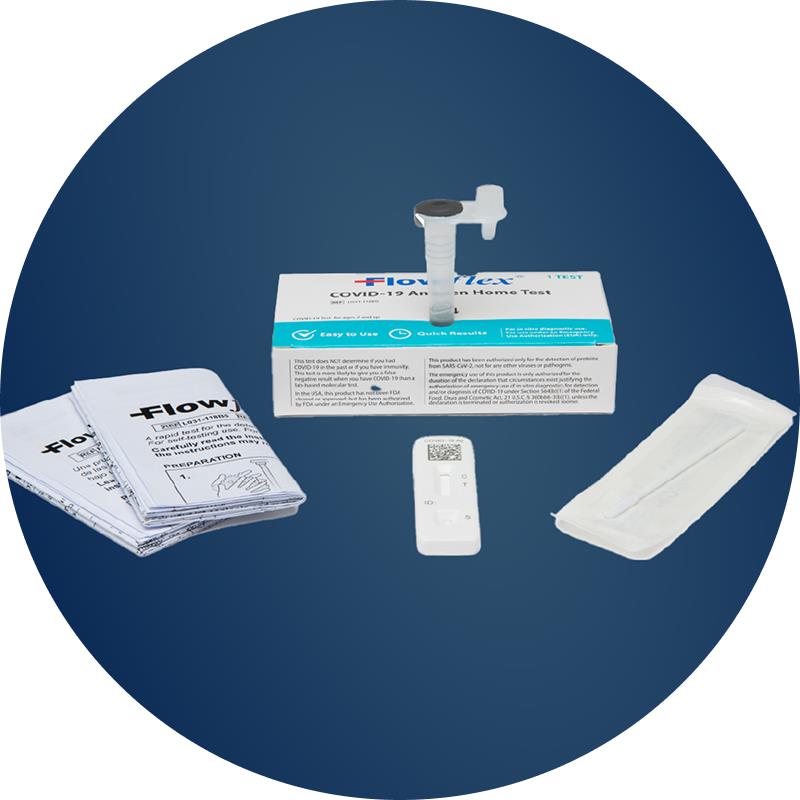
4 simple steps with clear instructions

Results in 15 minutes

Easy to carry anywhere you go

Safe for Children
Can be used to test children as young as 2 years old
No need to send off to a lab to obtain results
Compact packaging for “On-The-Go” testing
Easy-to-use nasal swab test
Results in 15 minutes
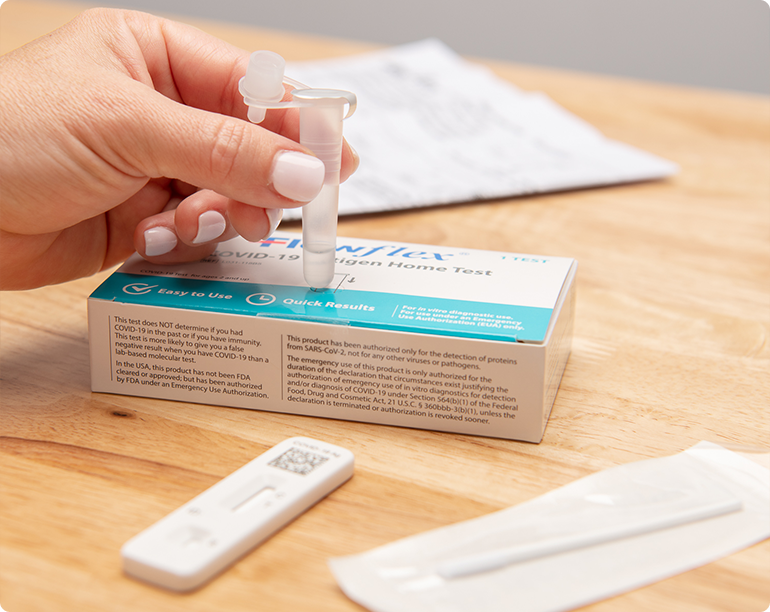
Easy to Use. Fast Results.
Please refer to the Package Insert for detailed instructions
Step 1
Swab
Step 2
Swirl
Step 3
Drop
Step 4
See Results
Easy Step-by-Step Video Tutorials
Frequently Asked Questions
Disclaimer
- In the USA, this product has not been FDA cleared or approved; but has been authorized by FDA under an EUA;
- This product has been authorized only for the detection of proteins from SARS- CoV-2, not for any other viruses or pathogens; and,
- The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
For more information on EUAs please visit: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization
For the most up to date information on COVID-19, please visit: www.cdc.gov/COVID19
Leading Technology
For more than two decades, ACON has led the way in making high quality diagnostic and medical devices more affordable to people all around the world.